Abstract
What is hunger? How do we know how much to eat? And what happens if we do not eat enough? To survive, the human body must have energy reserves available in the form of body fat. Using a hormone called leptin, the fat tissue regularly sends messages to the brain about the body’s energy reserves. When the brain receives a lot of leptin, it tells the body, “you are full, stop eating.” In certain situations, like obesity and pregnancy, the brain is less sensitive to leptin—so people do not feel full and they keep eating. We hypothesize that obesity and pregnancy change the blood vessels that transport leptin to the brain. As a result, an “illusion” of low energy reserves is created in the body, causing the brain to send fewer “stop eating” commands. As a result, people keep eating and gain weight.
Hormones Affect Whether We Feel Hungry or Full
People think about food a lot. How many times a day do you think about what you should eat—some chocolate, a snack, maybe a hot meal? Some of us might also wonder why it is hard to lose weight. Have you ever considered what it means to be “hungry” or “full,” and how these feelings relate to body weight? Have you ever wondered whether the body knows how much we should eat—and how it knows?
As humans, we communicate with others through writing, speaking, or using gestures. Inside the body, the organs communicate with each other with the help of substances called hormones. When an organ needs to pass a message to another organ, it releases a hormone into the blood—the type and amount of hormone carries the message. The blood transports the hormone throughout the body, and the organs that can absorb that hormone can understand its message. Each organ releases and absorbs several hormones. In this way, the organs have a lively conversation! The brain checks on the organs by paying attention to hormone levels, and it sends nerve signals or its own hormones to control what the others organs do and how we act.
Many hormones affect feelings of hunger and satiety (fullness). One of the most important ones is leptin. Leptin is a hormone released from the body’s fat cells that sends a message to the rest of the body about the level of body fat. Fat is the body’s energy storage [1]. Low levels of leptin tell the body that its energy reserves are depleting, and that it needs to start eating.
Leptin is absorbed by an area of the brain called the hypothalamus, which is the brain’s central control system. The hypothalamus affects growth, blood pressure, and the biological clock, as well as feelings of thirst and satiety. The hypothalamus affects the body by creating feelings of tiredness and hunger. In this way, the hypothalamus controls how much energy the body uses and takes in, helping to maintain normal body weight and ensuring a regular supply of energy to the body.
What Happens When the Body Does Not Respond to Leptin?
There are situations in which the hypothalamus does not properly receive the signal sent by the fat tissue via leptin, or it does not interpret that signal correctly. This state of decreased brain responsiveness to leptin is called leptin resistance [2]. In leptin resistance, the hypothalamus senses low levels of leptin, even though leptin levels are normal. When this happens, the hypothalamus “thinks” that the body does not have enough fat. In response, it decreases the rate of bodily processes and causes a feeling of hunger, which results in eating. Over time, the body will gain weight, and leptin levels will increase until the hypothalamus interprets them as normal.
Leptin resistance can be seen during pregnancy. During pregnancy, there is a decrease in the brain’s response to leptin, which leads the mother to eat more [3]. This is a good thing: during pregnancy, the body must supply nutrients and energy—not only to the mother but also to the growing fetus. Leptin resistance during pregnancy allows the mother to increase her fat reserves to serve the energy needs of the developing fetus, as well as to prepare for breastfeeding, which requires a lot of the mother’s energy (Figure 1).

- Figure 1 - During pregnancy, women often have increased feelings of hunger.
- One of the reasons this happens is because, during pregnancy, the body becomes resistant to leptin—the hormone that makes us feel “full” (Image created using DALL-E).
Basically, leptin resistance creates the “illusion” in the hypothalamus that the body does not have enough fat. Leptin resistance also occurs in obesity: although there is a large amount of fat tissue, the hypothalamus senses that the amount of fat is normal. Then, when the body loses weight, the hypothalamus interprets it as a drop below the optimum amount of fat, so it increases the bodily processes that cause weight gain, restoring obesity. The similarity between leptin resistance in pregnancy and obesity raises the question: Are the same mechanisms that cause increased eating during pregnancy active in obesity?
Scientists still do not fully understand what causes leptin resistance in pregnancy or obesity. This question is studied in many laboratories around the world because the growing problem of obesity is a worldwide concern. There are several main hypotheses: (1) maybe there are substances produced by the body that decrease leptin’s activity; (2) maybe stressful situations affect the body’s sensitivity to leptin; and (3) maybe the hypothalamus loses its ability to sense leptin. Our research investigates the third hypothesis.
How Does Leptin Get to the Brain?
How do hormones like leptin make their way to the brain? If you imagine how a hormone moves through the bloodstream and is absorbed by the brain, you might picture a ball floating down a river and reaching the shores of the brain, where it is plucked from the river by hard-working brain cells. However, the brain, unlike other organs, has almost no gentle riverbanks where hormones can arrive. Instead, the brain is a fortress, surrounded by a wall of steep cliffs that prevent hormones from entering. These “steep cliffs” are the brain’s blood vessels, which have unique properties that control the passage of substances into the brain from the blood stream, allowing necessary substances to pass while preventing the entry of any unwanted or dangerous substances. This system helps protect the brain from dangers like toxins and bacteria, and it is called the blood-brain barrier.
How does the hypothalamus sense hormone levels in the blood? Next to the hypothalamus, there is a tiny brain area called the median eminence, where the structure and function of blood vessels is unique [4]. The median eminence is like an opening in the cliff wall, through which hormones can enter and exit the brain via the bloodstream. Unlike the blood vessels in the rest of the brain, those in the median eminence are not sealed but instead look like tubes with tiny puncture holes. These holes are called fenestrae (from the Latin word for windows), and the diameter of each of them is <100 nm (one nm is one-millionth of a mm)—about a thousand times smaller than the thickness of a human hair [5]! Although it is important for sending and receiving hormone messages, the median eminence may also be a weak point in the hypothalamus. Any change in the median eminence—such as a decrease in the spacing, size, or number of fenestrae—will affect the ability of the hypothalamus to sense the levels of hormones in the blood and to release hormones into the bloodstream (Figure 2).
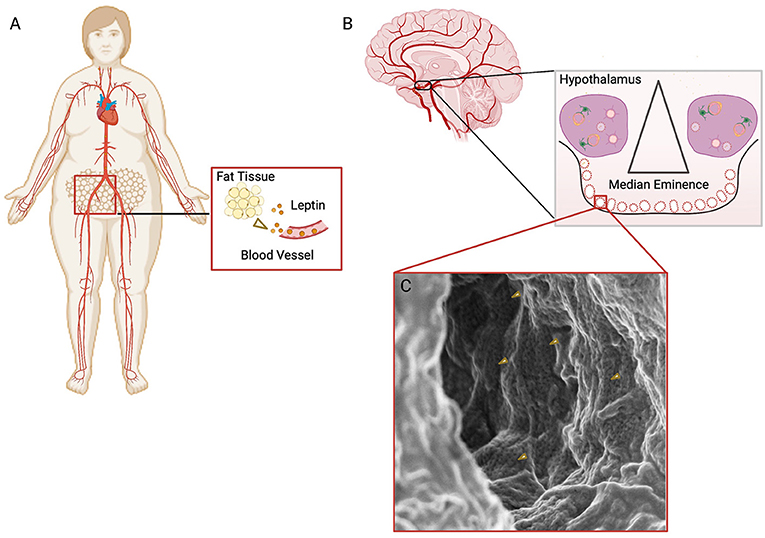
- Figure 2 - (A) Fat tissue releases leptin into the blood vessels, and the leptin travels through the bloodstream, eventually reaching the brain.
- (B) A brain structure near the hypothalamus, called the median eminence, contains fenestrated vessels through which leptin (yellow dots) enters the brain. (C) An electron microscope image of the inside of a fenestrated blood vessel in the median eminence. The dark dots are the fenestrae (some marked with yellow arrows), through which leptin can enter the brain.
Our Research
In our lab, we focus on two main questions: How does leptin get to the brain and hypothalamus? And, are there any obstacles leptin encounters on the way to the brain that could create leptin resistance? We hypothesize that part of the reason for leptin resistance might be that some of the fenestrae in the median eminence are closed. When there are not enough holes in the blood vessels of the median eminence for hormones to move through, not much leptin can reach the hypothalamus. This makes the brain think there is less leptin than there actually is. It is like a trick is being played on the brain.
But why would the fenestrae be closed? Something similar happens elsewhere in the body. Organs like the liver absorb and release proteins, hormones, and other substances through blood vessels. As people get older, the number of fenestrae in the liver decreases, and this affects the liver’s function [6]. We know that hormones can affect the number of fenestrae in the liver, and that the levels of some hormones change during pregnancy, obesity, and aging.
To investigate our hypothesis, we used a very powerful microscope called A super resolution microscope, which allows us to see the leptin molecules passing through the fenestrae. In doing so, we discovered that leptin, which is about 10 nm in diameter (one millionth the size of a human eyelash), not only passes through the fenestrae in the median eminence, but surprisingly moves through fenestrae in the hypothalamus as well (Figure 3).
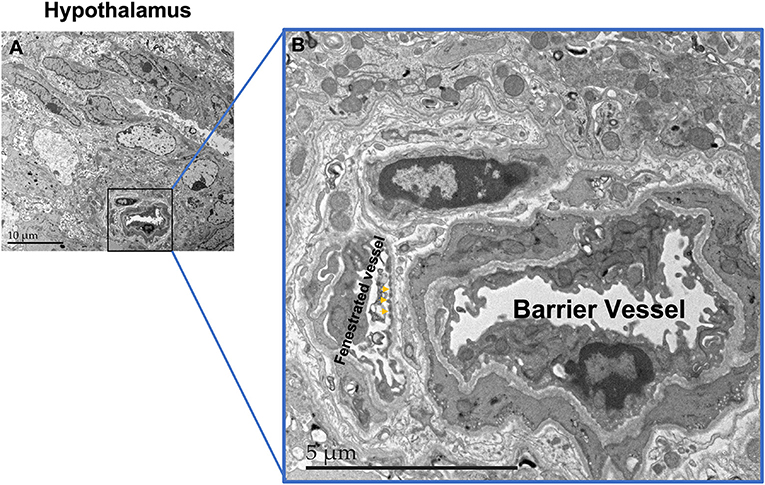
- Figure 3 - (A) Electron microscope image of the hypothalamus.
- The scale bar shows 10 micrometers (μm), with a micrometer being a millionth of a meter. (B) A zoomed-in image of the boxed area in A, showing two blood vessels: one is sealed and does not allow passage of substances (barrier vessel), and the other is fenestrated. Some of the fenestrae are marked with arrows.
We found that in pregnancy, fewer blood vessels in the median eminence are fenestrated, so there are fewer openings in total, and parts of the hypothalamus may not receive leptin at all. We also saw that the number of fenestrated blood vessels in the hypothalamus decreases during pregnancy, which reinforces the idea that less leptin reaches the brain in situations of leptin resistance, which can lead to misinterpreting the level of leptin in the brain and can result in overeating and weight gain.
Looking Ahead
Leptin entering the brain is crucial when discussing leptin resistance and weight gain. During pregnancy, changes in fenestrated capillaries support the idea that alterations in these capillaries may affect the entry of leptin into the brain. It is important to keep learning about the fenestrated vessels and researching the factors that affect the size and density of the fenestrae. This work will help us to better understand the communication that takes place between the body’s organs and the brain, through hormones. Specifically, this research can help us answer the question that concerns us all every day—to eat or not to eat?—as well questions about the right amounts of food for our bodies.
Glossary
Hormone: ↑ A substance released from an organ into the bloodstream that carries a biological message to distant organs.
Satiety: ↑ The feeling of being full and satisfied after eating enough to meet your body’s needs.
Leptin: ↑ A hormone released from fat cells that causes feelings of satiety when it is absorbed by the hypothalamus.
Hypothalamus: ↑ The area of the brain that controls basic aspects of the body’s activity such as hunger, thirst, blood pressure, the biological clock, and more.
Leptin Resistance: ↑ A condition in which the response of an organ to leptin decreases.
Blood-Brain Barrier: ↑ A “barrier” between the brain and the body created by properties of the brain’s blood vessels, which allow only certain substances to enter the brain as a protection mechanism.
Median Eminence: ↑ A brain structure near the hypothalamus where there are fenestrated blood vessels.
Fenestrae: ↑ Holes with a diameter of 50–100 nm (millionths of a millimeter) in blood vessels, which allow the passage of substances from and into the blood.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
[1] ↑ Friedman, J. M. 2019. Leptin and the endocrine control of energy balance. Nat. Metab. 1:754–64. doi: 10.1038/s42255-019-0095-y
[2] ↑ Izquierdo, A. G., Crujeiras, A. B., Casanueva, F. F., and Carreira, M. C. 2019. Leptin, obesity, and leptin resistance: where are we 25 years later? Nutrients 11:2704. doi: 10.3390/nu11112704
[3] ↑ Gustafson, P., Ladyman, S. R., and Brown, R. S. E. 2019. Suppression of leptin transport into the brain contributes to leptin resistance during pregnancy in the mouse. Endocrinology 160:880–90. doi: 10.1210/en.2018-01065
[4] ↑ Rodríguez, E. M., Blázquez, J. L., and Guerra, M. 2010. The design of barriers in the hypothalamus allows the median eminence and the arcuate nucleus to enjoy private milieus: the former opens to the portal blood and the latter to the cerebrospinal fluid. Peptides 31:757–76. doi: 10.1016/j.peptides.2010.01.003
[5] ↑ Stan, R. V. 2007. Endothelial stomatal and fenestral diaphragms in normal vessels and angiogenesis. J. Cell. Mol. Med. 11:621–43. doi: 10.1111/j.1582-4934.2007.00075.x
[6] ↑ May, D., Djonov, V., Zamir, G., Bala, M., Safadi, R., Sklair-Levy, M., et al. 2011. A transgenic model for conditional induction and rescue of portal hypertension reveals a role of VEGF-mediated regulation of sinusoidal fenestrations. PLoS ONE 6:e21478. doi: 10.1371/journal.pone.0021478
