Abstract
Our cells use the food we eat to produce the energy needed to do work. This amazing process is called metabolism and it is necessary for survival. If we eat more than we need, we will gain weight and may become obese. Obesity affects how cells and organs perform work and also increases the risk of metabolic diseases, which include type 2 diabetes and cardiovascular diseases. Metabolic diseases are the most common diseases of our time. National and global health authorities are working hard to prevent and treat obesity and obesity-related diseases. However, some people with obesity appear to have normal metabolism. Why do some obese people develop metabolic diseases while others do not? In this article, we discuss how metabolism may differ in people with obesity. We also describe some key reasons why some people with obesity may be healthier than others.
Metabolic Regulation Is Necessary for Survival
The work that the cells of an organism perform to stay alive is called metabolism. The energy required for metabolism comes from nutrients in foods that we eat, digest, and absorb into the bloodstream (Figure 1). Carbohydrates, fats, and proteins from foods are energy sources for the body’s cells. A healthy diet—including fruits and vegetables, whole grains, and fish—is an important source of nutrients and energy. Together with exercise, a healthy diet will result in a well-regulated metabolism and a reduced risk of certain diseases.
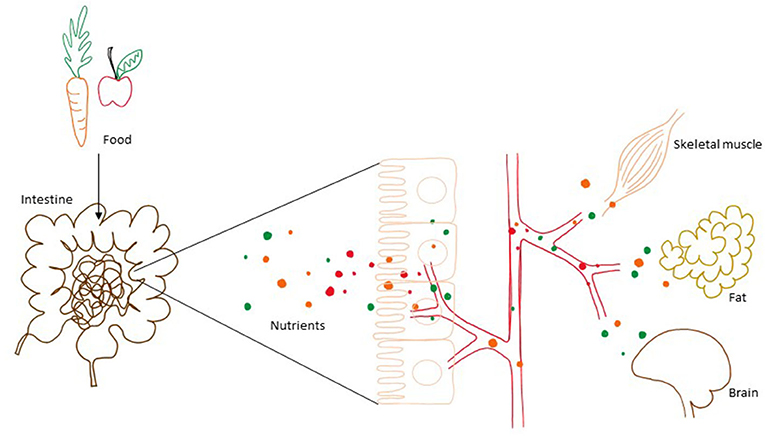
- Figure 1 - Food is digested in the gut and nutrients, like glucose and fat, are taken up by the blood.
- The blood transports the nutrients to cells and organs, which use them for energy metabolism. If energy is not needed, the nutrients are converted to fat and stored for later use.
Carbohydrates are the most important energy source for the body’s cells. Carbohydrates are taken up into the blood from the intestine, in the form of a sugar called glucose. The level of glucose in the blood increases after a meal and decreases again when the cells take it up. To fuel our bodies when we are not eating, we also need to store metabolic fuels. Fat is the most energy-dense nutrient and is the form in which energy is stored when we take in more food than we currently need for energy.
Metabolism must be carefully regulated so that our bodies can deal with short- and long-term changes in energy availability. This regulation involves communication between various tissues and organs. After a meal, when we start to exercise, or after an overnight fast, the body’s organs must be informed about whether energy should be stored or mobilized from storage sites. Much of this communication is done by signaling molecules traveling in the blood, including hormones and metabolites (chemical products of metabolism). The body’s ability to store energy as fat is important for survival and allows us to vary our energy intake from day to day. Today, however, most of us live inactive lives and have an abundance of tasty and energy-dense foods available. Unfortunately, this makes it easy to eat more food than the body needs, leading to increased weight and obesity.
Obesity Is Elevated Weight Relative to Height
All over the world, the number of people with obesity has been increasing [1]. The number of children with obesity is also increasing, and during the last 40 years (1975–2016) the number of girls with obesity has increased from 5 to 50 million and the number of boys with obesity has increased 6–74 million [2]. But what is obesity? Obesity is simply when a person’s weight is elevated relative to their height. To figure out how elevated a person’s weight must be to be considered obese, we must calculate the body mass index, or BMI. BMI is the weight in kilograms divided by the square of the height in meters (kg/m2). Using this measure, people fall into one of four categories: underweight, normal weight, overweight, and obesity. Obesity is defined as a BMI of 30 kg/m2 or more. Note that, by this definition, a person defined as obese may have a lot of muscle mass. However, most people with obesity have large body-fat stores.
Obesity Increases the Risk of Metabolic Diseases
There are two main stores of body fat: underneath the skin, called subcutaneous fat, and surrounding the organs in the abdomen, called visceral fat. The visceral fat stores are the most important for metabolic regulation—if a person’s visceral fat stores increase, at some point, their metabolic regulation will be affected and may even be dysfunctional. Over time, a dysfunctional metabolism can lead to metabolic diseases such as type 2 diabetes and cardiovascular diseases [3]. These are the most common diseases of our time, and both national and global health authorities are working to treat and prevent obesity and obesity-related diseases.
Weight reduction will normally improve metabolic regulation and thereby reduce the risk of metabolic diseases. But unfortunately, weight reduction may be difficult to maintain for a long period—often excess weight is regained. To effectively prevent and treat obesity-related diseases, we need more knowledge about how metabolism is affected by obesity.
Biomarkers of Health and Disease
If you have an infection, you often know this because you feel sick and have a fever. Metabolic diseases take years to develop, and people often do not have obvious symptoms while these diseases are developing. To understand a person’s risk for metabolic diseases, blood samples are taken to measure biomarkers—substances that indicate the status of metabolism and overall health.
The amounts of glucose and fat in the blood are carefully regulated as part of normal metabolism. In addition, molecules called fat transporters function to move fat throughout the body via the blood. When fat stores increase, so do the amounts of glucose, fat, and fat transporters in the blood. These substances can serve as biomarkers and their levels are therefore used as indicators of health status. There may be many other biomarkers of metabolic regulation that may be discovered as we continue to study the metabolism of people with obesity compared to people with normal weight. Maybe there are markers that can warn us about the risk of metabolic diseases long before they occur!
People With Obesity Are Diverse
Although obesity generally increases the risk of developing metabolic diseases, people with obesity are diverse. Some people with obesity are described as low-risk obesity meaning, when compared to people with high-risk obesity, they have healthier blood fat levels and normal or only slightly affected blood glucose regulation, despite having similar amounts of body fat [4, 5]. Why is this? Could there be undiscovered biomarkers that explain this difference in obesity-related metabolic regulation? Perhaps individuals with low-risk obesity will transition to high-risk obesity with time?
One way to start answering these questions is to study metabolic regulation in people with low-risk and high-risk obesity—and that is what we did. We created a detailed map of many kinds of molecules in the blood that may increase our understanding of metabolic regulation in obesity, to see whether any of those molecules might serve as new biomarkers of health and disease.
Characteristics of Individuals With Low-Risk Obesity
Our study included 30 participants (18 males and 12 females) who were divided into three groups: low-risk obesity, high-risk obesity, and healthy normal weight. Participants were grouped based on BMI and blood levels of fat and glucose. While the low-risk obesity and normal-weight groups had normal levels of fat and glucose in blood, the high-risk obesity group had high blood levels of these biomarkers. In the low-risk obesity group, there were nine participants with a mean age of 49 years and mean BMI of 33 kg/m2. The high-risk obesity group had 10 participants with a mean age of 52 years and mean BMI of 32 kg/m2. The normal weight group had 11 participants with a mean age of 47 years and mean BMI of 23 kg/m2 [6]. To investigate possible new biomarkers for high- and low-risk obesity, we analyzed blood samples for a wide range of metabolites.
We found that the low-risk obesity group had lower levels of various fat transporters in their blood compared to the high-risk obesity group (Figure 2). They also had lower levels of some amino acids—the building blocks of proteins—namely isoleucine, leucine, and valine. The normal-weight group had lower levels of the amino acid phenylalanine compared with the low-risk obesity group. The types of fat found in the blood were more favorable, meaning less of the fat that cause cardiovascular disease, in the low-risk obesity group compared with the high-risk obesity group (Figure 2).
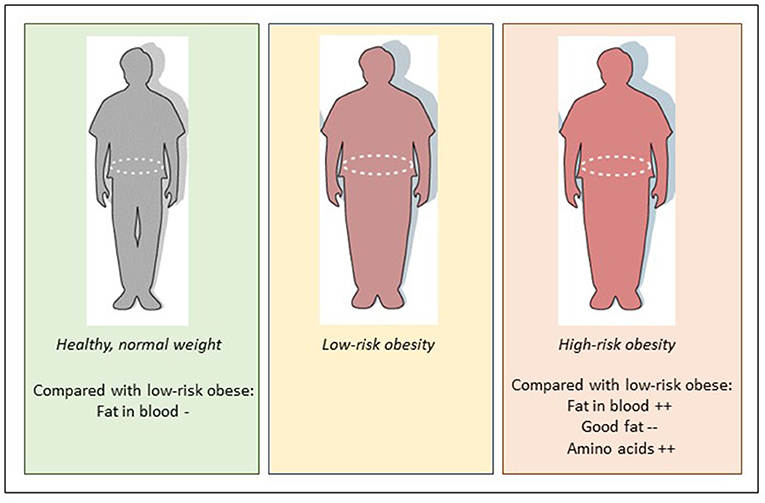
- Figure 2 - Compared to low-risk obesity, high-risk obesity is characterized by higher blood levels of fat and amino acids, and lower blood levels of “good fat”.
- Compared to low-risk obesity, normal weight is characterized by lower blood levels of fat and unchanged levels of amino acids.
The low-risk obesity profile could possibly be explained by a healthier lifestyle. Although our study did not completely rule out a role of diet, we found no differences in dietary intake of total energy, fat, protein, carbohydrates, or alcohol between people with low-risk obesity and high-risk obesity. In this study we did not measure the participants’ regular physical activity level. Physical activity is beneficial for metabolic regulation and might explain some of the differences between the two obesity groups. There may also be genetic variation between the groups explaining the differences. Although we do not yet understand why some people with obesity do not have the same risk of developing metabolic diseases, it is possible that these individuals will have metabolic diseases at a later timepoint. This means that obesity related metabolic diseases may be delayed or even prevented if we know the biomarkers that reflect the risk at an earlier timepoint.
Conclusion
People with obesity are diverse—some develop high-risk obesity, while others are more fortunate. Our study found that low-risk obesity is characterized not only by levels of blood glucose and fats in the normal range, but also by other metabolic markers, such as lower circulating levels of amino acids and a broad range of fats. This might mean that some biomarkers may be more predictive of metabolic disease risk. These findings may help to provide a deeper understanding of obesity and its complications, which may eventually improve prevention and treatment of obesity and health.
Acknowledgments
This work was funded by the Throne Holst Foundation for Nutrition Research, Oslo, Norway; University of Oslo, Oslo, Norway, OsloMet, Oslo, Norway; and Mills DA, Oslo, Norway. We are grateful to all participants in the study, to researchers Nima Wesseltoft-Rao and Ingunn Narverud, and to bio-engineer Ellen Raael for their contributions to the present study. The study protocol and statistical analysis plan is available by request to smulven@medisin.uio.no.
Glossary
Metabolism: ↑ All the work that cells of a living organism perform to stay alive.
Glucose: ↑ A simple sugar molecule (a monosaccharide, a type of carbohydrate). An important energy source for cells, and always found in blood.
Obesity: ↑ A body mass index (BMI) of 30 kg/m2 or more.
Body Mass Index: ↑ A measure of excess adiposity. Calculated as a person’s weight in kilograms divided by the square of the height in meters: kg/m2.
Metabolic Diseases: ↑ An umbrella term for a broad range of diseases affecting metabolism, including obesity, type 2 diabetes, cardiovascular disease.
Type 2 Diabetes: ↑ A condition in which cells cannot use blood glucose efficiently for energy, so the glucose level in blood gradually gets too high.
Cardiovascular Disease: ↑ A general term for conditions affecting the heart or blood vessels, such as heart attack and stroke.
Biomarker: ↑ A substance that is measured in blood, urine, or another tissue and is used as an indicator of normal or abnormal biological processes.
Conflict of Interest
Mills AS partially funded the study and VT-H has been employed at Mills AS. She does not own any stocks in the company, and the work performed in this paper was done after she left the company. VT-H collaborates with and/or has received research grant from Mills AS, Det Glutenfrie Verksted, Mesterbakeren and the Norwegian Celiac Disease Association. KH has received research grant from TINE BA, Olympic Seafood, Amgen, Sanofi, Kaneka and Pronova. SU has received research grant from TINE BA and Olympic Seafood. None of these grants or honoraria are related to the content of this manuscript.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Original Source Article
↑Telle-Hansen, V. H., Christensen, J. J., Formo, G. A., Holven, K. B., and Ulven, S. M. 2020. Acomprehensive metabolic profiling of the metabolically healthy obesity phenotype. Lipids Health Dis. 19:90. doi: 10.1186/s12944-020-01273-z
References
[1] ↑ Collaboration NCDRF. 2016. Trends in adult body-mass index in 200 countries from 1975 to 2014: a pooled analysis of 1698 population-based measurement studies with 19.2 million participants. Lancet. 387:1377–96. doi: 10.1016/S0140-6736(16)30054-X
[2] ↑ Collaboration NCDRF. 2017. Worldwide trends in body-mass index, underweight, overweight, and obesity from 1975 to 2016: a pooled analysis of 2416 population-based measurement studies in 128.9 million children, adolescents, and adults. Lancet. 390:2627–42. doi: 10.1016/S0140-6736(17)32129-3
[3] ↑ Collaborators GBDO, Afshin, A., Forouzanfar, M. H., Reitsma, M. B., Sur, P., Estep, K., et al. 2017. Health effects of overweight and obesity in 195 countries over 25 years. N. Engl. J. Med. 377:13–27. doi: 10.1056/NEJMoa1614362
[4] ↑ Munoz-Garach, A., Cornejo-Pareja, I., and Tinahones, F. J. 2016. Does metabolically healthy obesity exist? Nutrients. 8:320. doi: 10.3390/nu8060320
[5] ↑ Karelis, A. D., Brochu, M., and Rabasa-Lhoret, R. 2004. Can we identify metabolically healthy but obese individuals (MHO)? Diabetes Metab. 30:569–72. doi: 10.1016/S1262-3636(07)70156-8
[6] ↑ Telle-Hansen, V. H., Christensen, J. J., Formo, G. A., Holven, K. B., and Ulven, S. M. 2020. A comprehensive metabolic profiling of the metabolically healthy obesity phenotype. Lipids Health Dis. 19:90. doi: 10.1186/s12944-020-01273-z
