Abstract
Everything we do (sense, move, feel, remember…) relies on the activity of neurons in the brain, the connections between them and their ability to change the strength of these connections. Astrocytes are a different type of brain cells previously thought to merely support the activity of neurons. Advanced tools allow us to artificially activate specific cells populations in specific brain areas. Using such tools, we found that astrocytes can independently strengthen the connections between neurons and improve memory. We show that this memory improvement is due to the ability of astrocytes to enhance only the activity of those neurons participating in learning. Our findings suggest that astrocytes are not merely support cells enabling memory, but they can actively change the function of neurons. The ability of astrocytes to enhance memory may serve as a basis for future treatments to improve normal memory or help people who suffer from memory loss.
Star Cells in the Brain
Nerve cells (neurons) are the best known cell population of the brain. They respond to stimuli, communicate with each other, perform computations, and transfer information over long distances. The communication between neurons allows us to sense, think, talk, feel, and move. In fact, almost every feature that we possess, no matter how complicated, begins in our nerve cells. Mental activity, similar to processes like vision or hearing, is represented in our brains by a group of cells communicating with one another. The difference between one process and another, such as the difference between seeing and hearing, depends on which neurons are active. The ability of neurons to change the strength of the connections between them defines the flexibility level of their communication, and it is thought to be critical for information processing and memory formation. However, neurons are not alone (they are not even the majority) in the brain. Several types of glial cells (glia means glue in Greek) surround and support the neurons.
A major type of glia cells are astrocytes, which means “star cells” in Greek, called so for their shape (Figure 1). Astrocytes are in close contact with multiple nerve cells in the brain (Figure 2A), and can sense the activity of their surrounding neurons and respond to changes in the environment. This sensitivity allows astrocytes to provide nerve cells with more energy when they are active, remove waste materials, and release essential growth factors to help the neurons grow. In addition to these supportive roles, astrocytes can monitor and directly change neuronal activity. Several pioneering studies have shown astrocytes to be necessary for changing neuronal communication, and therefore for normal memory function [1].
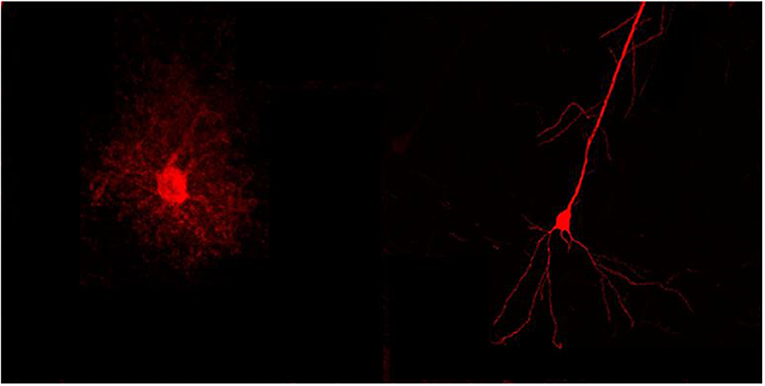
- Figure 1 - Nerve cell vs. astrocyte.
- A nerve cell (right) differs from an astrocyte (left) by its structure, among other things. The nerve cell is characterized by long, thin processes emerging from the cell body (circular part), that rarely branch. The astrocyte is characterized by a bushier structure, where every branch splits multiple times.
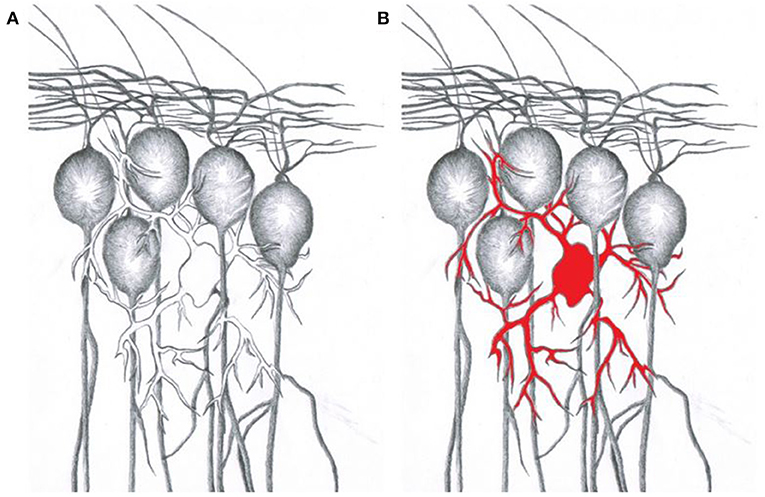
- Figure 2 - The star cells of the brain.
- (A) Astrocytes (“star cells,” shown in white) are in close contact with their surrounding neurons (gray). (B) Using genetic engineering, proteins allowing modulation by drugs or light (red) can be specifically expressed in astrocytes, but not in their neighboring neurons.
Memory and Its Enhancement
Our memories guide our present and future behaviors based on our past experiences. They define who we are and how we experience the world. During learning, in a process called memory allocation, a group of active neurons are selected to serve as the cells that “hold” a specific memory. These cells will tend to be active together from that time forward, so that each time this memory is recalled, this chosen group of neurons will be reactivated [2]. Therefore, recalling a memory depends on the reactivation of the same group of cells that were activated at the time the memory was made.
Memory disruption is relatively easy to induce in the lab, but what most of us really want, even if our memory is perfectly normal, is to make it even better. This aspiration for memory improvement has challenged scientists for many years. Most attempts to improve memory focus on strengthening the connections between neurons or boosting the formation of memories [2, 3].
Is it possible that astrocytes can sense the neuronal activity around them and use this information to improve the flexibility of neuronal communication and possibly improve memory?
How Do We Study the Role of Astrocytes in Memory?
Many studies have demonstrated the necessity of astrocytes for the flexibility of neuronal communication and for memory, by showing that interrupting the activity of astrocytes leads to problems with these functions. However, the methods used for these experiments were either extremely prolonged (removing part of the brain, for example), or not entirely specific to astrocytes. To further study the ability of astrocytes to improve memory in behaving animals, one needs a method to artificially control astrocyte activity specifically, quickly, and in a reversible way. Recently, genetic engineering techniques allowing such precise experiments were developed for neurons, and these techniques can be similarly used in astrocytes (Figure 2B). One technique, called chemogenetics, allows researchers to turn a specific cell population sensitive to a drug that the cells are usually not sensitive to. Another technique, called optogenetics, can similarly turn a specific population of cells sensitive to light. For example, we can specifically activate astrocytes, without affecting neurons or any other type of cells in the brain, using chemogenetics, in a fast (within minutes) and reversible (gone within hours) way. Similarly, we can choose to activate neurons but not astrocytes.
Using these tools, we investigated whether astrocytic activity is not only necessary, but also sufficient to induce changes in neuronal communication and improve memory [4].
Activating Astrocytes for Memory Improvement
When we activated astrocytes in the hippocampus, which is the brain region responsible for memory, we found that they can independently increase the strength of connections between neurons in this region (Figure 3A) [4]. It seems that astrocytes are not only necessary for the ability of neurons to strengthen their connections, but that their activation can actually cause this change in neurons.
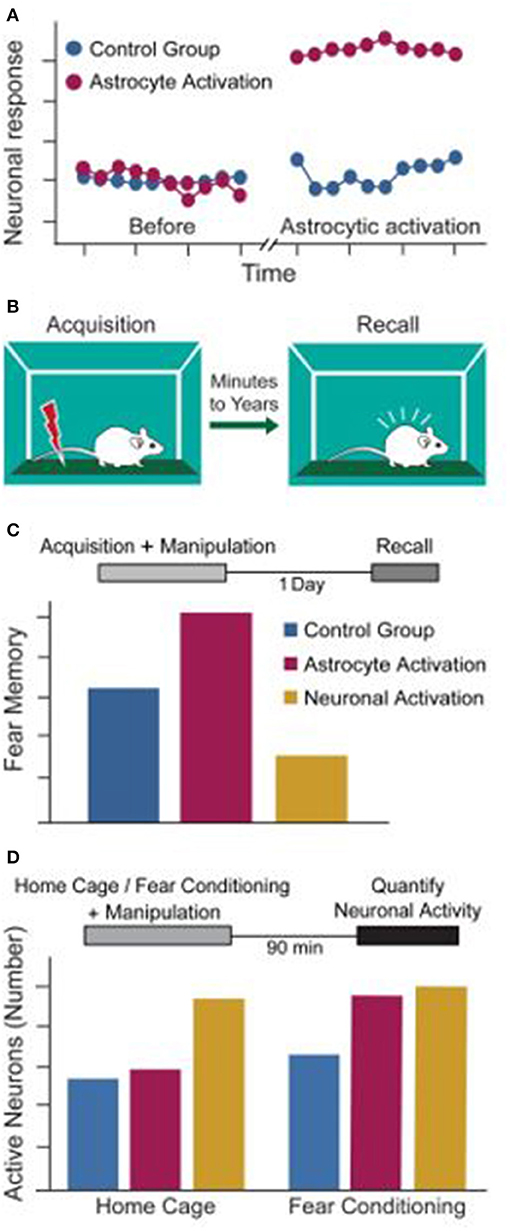
- Figure 3 - Astrocytes for memory improvement.
- (A) Astrocytic activation increases the strength of connections between neurons and causes an improved neuronal response. (B) Memory in mice can be measured by fear conditioning. In this method, mice learn to associate a new location with an electrical foot-shock. When re-exposed to this environment, the mice will show fear, even though no shock is present at that time. (C) Astrocytic activation (crimson) during memory acquisition improves fear memory recall on the next day, whereas neuronal activation (yellow) impairs retrieval, compared to a control group (blue). (D) Astrocytic activation modulated neuronal activity in a task-dependent way. Specifically, it increases the number of active neurons only when coupled with fear conditioning learning. Direct activation of neurons increases the number of active neurons whether learning is occurring or not.
Based on this surprising finding, we then tested whether astrocytes can also improve memory performance. In a behavioral paradigm call “fear conditioning” we taught mice to recognize a specific environment and associate it with fearful event. We then tested their memories the next day (Figure 3B). The astrocytes were activated either during learning or during recall, to test the involvement of these cells in memory formation and recall. Activating astrocytes during learning dramatically improved memory the next day (Figure 3C), whereas astrocytic activation during recall had no effect [4]. These findings, which were recently replicated by another group [5], suggest that astrocytes can improve memory acquisition, but they do not improve memory retrieval.
Our findings that astrocytic activation strengthened neuronal connectivity and improved memory raises the tempting hypothesis that astrocytes react to the neuronal activity around them and regulate this neuronal activity in a meaningful way, leading to improved memory. Alternatively, memory improvement could simply be caused by a general increase in neuronal activity stimulated by the astrocytes, suggesting they play no “smart” role in memory. To test the latter possibility, we directly activated neurons (instead of astrocytes) in the hippocampus. Neuronal activation during learning did not improve memory (Figure 2C). On the contrary, it induced a severe memory impairment [4]. This suggests that a general increase in neuronal activity cannot explain the memory improvement caused by astrocytes, but rather that astrocytes cells play a more sophisticated role in the learning process.
Astrocytes Affect Neuronanl Activity When Coupled With Learning
Why does astrocytic activation improve memory while neuronal activation impairs it? Previous research had shown that a small group of neurons active during learning are more likely to be part of the ensemble supporting this memory. Such ensembles are later necessary for recall, and increasing the activity of this small neuronal population before training can improve memory [2, 3].
To show that the memory improvement is caused by astrocytes and it is not merely due to the general increase in neuronal activity, but rather as a response of astrocytes to the activity of their surrounding neurons, we performed the following experiment: we activated either astrocytes or neurons in two groups of mice—one group that was exposed to a new location to acquire a new memory, and another group that just stayed in their familiar home-cage and did not develop a new memory. We then measured the number of active neurons in each group of mice. Activation of astrocytes increased the activity of neurons only when coupled with learning, but not in home-caged mice (Figure 3D). Neuronal activation increased the number of active neurons regardless of whether a new memory was being made or not [4].
Our results show that astrocytic activation increases the activation of neurons in a task-dependent way, only when coupled with learning, leading to improved memory allocation. Directly activating neurons, on the other hand, causes a non-selective increase in activity regardless of external experiences, which impairs the distinction between important and irrelevant activity, and impairs memory.
Conclusions and Future Potential
Our findings that astrocytes can induce flexibility with the communication among the neurons and improve memory suggests that astrocytes have the ability to independently compute task-specific information based on their surrounding neuronal activity, and use it to optimally modulate the neuronal circuit. In other words, astrocytes are not just support cells enabling changes in neuronal connections and cognitive function, but in fact play an active role—they can actively change and improve the function of the networks of neurons that are responsible for our thoughts, emotions, and perceptions.
The ability of astrocytes to augment memory acquisition may have important clinical implications for cognitive improvement treatments. The major advantage of working with astrocytic to improve memory is that the astrocytes act specifically on the active neurons, they do not generally activate all neurons in the brain. This specificity might help scientists to develop drugs to help improve memory without activating other brain cells that are not involved in the memory-making process.
Glossary
Neuronal Communication: ↑ The transfer of information between one neuron to another in a network. Each neuron in the brain can potentially interact with thousands of other neurons, and the strength of their connections will determine how significantly will they influence one another.
Memory Allocation: ↑ The process determining which specific neurons will represent a given memory.
Chemogenetic: ↑ The use of artificial receptors activated by otherwise inert drugs, to modulate the activity of selected cells.
Optogenetic: ↑ The use of light-activated proteins to control selected cells.
Cognitive: ↑ The mental process of learning from perception, thought and comparison to previous experience.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Original Source Article
↑Adamsky, A., Kol, A., Kreisel, T., Doron, A., Ozeri-Engelhard, N., Melcer, T., et al. 2018. Astrocytic activation generates de novo neuronal potentiation and memory enhancement. Cell 174:59–71.e14. doi: 10.1016/j.cell.2018.05.002
References
[1] ↑ Araque, A., Carmignoto, G., Haydon, P. G., Oliet, S. H., Robitaille, R., and Volterra, A. 2014. Gliotransmitters travel in time and space. Neuron 81:728–39. doi: 10.1016/j.neuron.2014.02.007
[2] ↑ Josselyn, S. A., Kohler, S., and Frankland, P. W. 2015. Finding the engram. Nat. Rev. Neurosci. 16:521–34. doi: 10.1038/nrn4000
[3] ↑ Lee, Y. S., and Silva, A. J. 2009. The molecular and cellular biology of enhanced cognition. Nat. Rev. Neurosci. 10:126–40. doi: 10.1038/nrn2572
[4] ↑ Adamsky, A., Kol, A., Kreisel, T., Doron, A., Ozeri-Engelhard, N., Melcer, T., et al. 2018. Astrocytic activation generates de novo neuronal potentiation and memory enhancement. Cell 174:59–71.e14. doi: 10.1016/j.cell.2018.05.002
[5] ↑ Mederos, S., Hernández-Vivanco, A., Ramírez-Franco, J., Martín-Fernández, M., Navarrete, M., Yang, A., et al. 2019. Melanopsin for precise optogenetic activation of astrocyte-neuron networks. Glia 67:915–34. doi: 10.1002/glia.23580
