Abstract
When a female crested newt lays her clutch of about three-hundred eggs, half of these will never hatch. What an incredible waste! It turns out that these newts suffer from a deadly hereditary disease called a balanced lethal system. Here is how it works: in a balanced lethal system, there are two distinct versions of a particular chromosome. Newts need both to live. However, which version a fertilized newt egg gets from its father and mother is random. Therefore, there is a 50% chance that it receives the same version twice—and misses the other half of the critical pair. Hence, half of the eggs end up with the wrong chromosome combination and die. The newts are not the only example; balanced lethal systems are found in some plants and insects as well. Why something so disadvantageous as a balanced lethal system would ever evolve is still a big, evolutionary mystery!
Unfortunate Eggs
Two centuries ago, Mauro Rusconi was studying the eggs of crested newts. The nice thing about crested newt eggs is that they are big and transparent, so you can easily see how the embryo inside of them develops. Mauro was struck by how many of the little newt embryos die during development. In fact, exactly 50% of all eggs die before hatching (Figure 1). He described his findings in his book Amours des Salamandres Aquatiques (The Love Life of Newts), published in 1821 [1]. Since then, Mauro’s observations have been confirmed time and again, in all the species of crested newt, as well as their close cousins: the marbled newts. With his grim discovery, Mauro stumbled upon one of the greatest mysteries in evolution. How is it possible that something so wasteful could ever evolve?

- Figure 1 - Half of the eggs of crested newts never hatch! (A) During the breeding season, male crested newts (left) display a splendid ornamental crest (hence the name) to seduce the females (right).
- After mating, the female carefully wraps each of her several 100 eggs in a separate leaf for protection. (B) The weirdest thing happens when these eggs develop, as can be seen in a time-series of pictures taken for two eggs. While half of the eggs develop normally and hatch (left), the other half simply stop halfway and die (right).
Strange Chromosomes
For a long time, no one understood why so many newt eggs were dying. Scientists gradually gained a better understanding when they started studying the chromosomes of crested newts [2]. In most animals, including newts, chromosomes come in pairs. Crested newts have 12 pairs of chromosomes—so, 24 chromosomes in total. When a newt produces sex cells, each sperm or egg cell receives a single chromosome from each pair, so sex cells contain only half the normal number of chromosomes. During fertilization, a sperm and an egg cell merge. This way, the fertilized egg obtains a new set of chromosome pairs, each pair with one chromosome from its father and one from its mother, and the normal chromosome number is restored. In crested newts, something strange happens when one of the chromosome pairs is inherited.
The first clue arrived when researchers realized that the longest pair of chromosomes, chromosome 1, behaves rather differently compared to the other 11 pairs. Normally, before new sperm or egg cells are produced, the two chromosomes of each pair are kind of shuffled together—they exchange chunks of genetic material and then split up. This means that each sperm or egg cell obtains a single set of chromosomes that have a new and unique combination of DNA. This process is called recombination, and it is important because it allows broken pieces of DNA to be removed without throwing away an entire chromosome. However, in crested newts, chromosome 1 does not get shuffled like this. This means that, in each sperm or egg cell, a newt passes on a copy of chromosome 1 that is exactly the same as the one it inherited from its mother or father, but never a mixture of both (Figure 2).
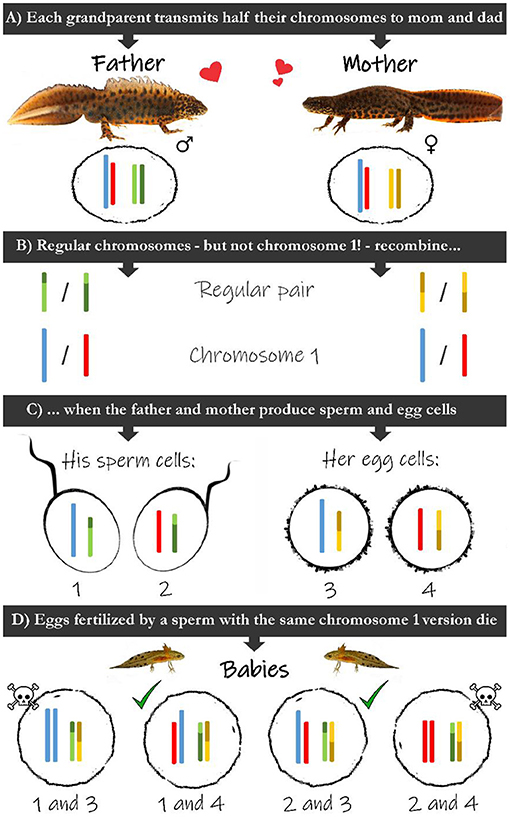
- Figure 2 - (A) Fathers and mothers have pairs of chromosomes for which they obtained one each from their parents.
- (B) When sperm or egg cells are produced, a regular chromosome pair recombines, meaning it shuffles blocks of DNA. In contrast, the two versions of chromosome 1 do not recombine. (C) Each sex cell receives a single chromosome for each pair: a regular chromosome will have been recombined, while the chromosome 1 version is passed on unaltered. (D) Upon fertilization chromosomes pair up again. Only those fertilized eggs that receive the two different versions of chromosome 1 can survive.
The second clue was that chromosome 1 actually comes in two versions that look different from each other under the microscope. We now call these two versions chromosome 1A and 1B. 1A is slightly longer than 1B. Having two versions of a chromosome is not that unusual. Lots of organisms have a chromosome pair called the sex chromosomes, in which the individual chromosomes may differ in shape. These chromosomes influence what sex the animal is: the males have two different versions of the chromosome pair (called X and Y in humans, for example), while the females have two identical ones (two Xs in humans), or the other way around (in birds, for example, it is the males who have two identical sex chromosomes). For a while, chromosome 1 was thought to determine the sex of crested newts. However, several scientists noticed that all crested newts, including both males and females, have both the 1A and 1B version. Therefore, chromosome 1 probably had nothing to do with sex. In fact, it later turned out that chromosome 4 controls the sex of crested newts. So, what is going on with that strange chromosome 1?
Balanced Lethal System
In 1980, over 150 years after Rusconi’s initial observations, Herbert Macgregor and Heather Horner finally discovered that there is a connection between this weird chromosome 1 and the grim fate of half the crested newt eggs [3]. Remember that adult crested newts only put a single chromosome from each pair into each of their sperm or egg cells. This means they can either transmit chromosome 1A or chromosome 1B in each sex cell. In a fertilized newt egg, the single chromosomes in the sperm and egg cell are brought together in pairs again. We know that all adult newts carry both 1A and 1B, so they either got 1A from mother’s egg cell and 1B from father’s sperm cell, or the other way around.
However, those are not the only combinations possible! What happens if an egg cell with 1A gets fertilized by a sperm cell that also has 1A? Or if an embryo inherits chromosome 1B from both its parents? It turns out that these two chromosome combinations, 1A+1A and 1B+1B, are deadly! The fertilized eggs with these chromosome combinations are the ones that do not hatch (Figure 2). Apparently, a crested newt needs a copy of both 1A and 1B to survive. This suggests that 1A has some error that that is not present on 1B, and vice versa. Normally such errors can be fixed by recombination; chromosomes can swap the faulty DNA for functional DNA. However, remember that, in crested newts, there is no recombination in chromosome 1. This means that 1A and 1B are stuck with their particular errors, which is why 1A is required to fix the error on 1B, and 1B is needed to fix the error on 1A!
Herbert and Heather realized that their findings neatly explained why exactly 50% of the crested newt eggs are doomed. Because 1A or 1B end up in a sex cell at random, on average half of sperm or egg cells will possess 1A and the other half 1B. This means that, for each fertilized egg, there is a 50% chance the sperm cell will carry the same version of chromosome 1 as the egg cell. In this case, the fertilized egg will die (Figure 3). Since all the eggs that survive and hatch must have both 1A and 1B, when these eggs grow into adults, they will again produce sperm and egg cells with the same mix of 1A and 1B as their parents did. Consequently, the loss of half the eggs keeps occurring, every single generation. Such a situation is known as a balanced lethal system [4]. Basically, a balanced lethal system is a deadly hereditary (inherited) disease.
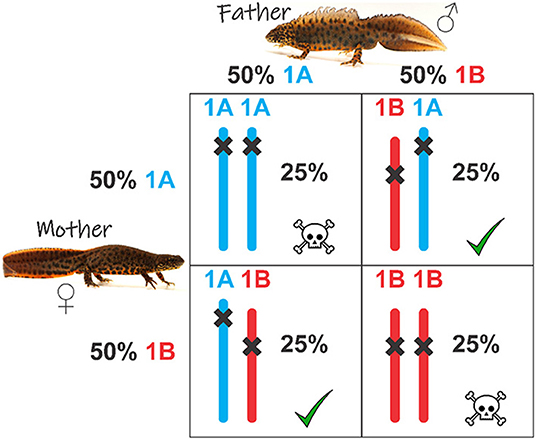
- Figure 3 - The 4-panel box shows the possible fertilized eggs produced by the parents, who each have both a 1A and a 1B version of chromosome 1.
- 1A and 1B have unique errors (shown with “x”). Parents transmit either 1A or 1B to each sex cell, with a 50% chance. Therefore, four combinations of fertilized eggs are possible, each with a 25% chance. In eggs with a 1B+1A or 1A+1B combinations, the errors in each version of chromosome 1 are compensated by the other version. This is not the case in eggs that contain the 1A+1A or 1B+1B combinations, causing these eggs to die.
An Evolutionary Puzzle
Balanced lethal systems are just bizarre. Evolution by natural selection is supposed to maximize the number of offspring produced by parents. However, in a balanced lethal system, half of every generation dies! Newts that do not suffer from a balanced lethal system could have twice as many offspring. You would think it should be impossible for this wasteful situation to evolve—balanced lethal systems seem to go against evolution! More surprising is that balanced lethal systems do not only occur in crested newts. They have also been described in species very different from newts, including plants and insects. So, despite their obvious disadvantage, balanced lethal systems appear to be a recurring feature of evolution. Why would that be so? This is quite the puzzle!
How is it possible that these disadvantageous balanced lethal systems keep evolving? Well, evolution by natural selection is not an “intelligent” process—it just happens! And while evolution can create brilliant adaptations, there is no planning ahead. This means that some feature that provide individuals with a short-term benefit would automatically spread through the population by natural selection, even though that feature might turn out to be disadvantageous for the entire species in the long run. For example, many bird species whose ancestors found their way to remote islands can no longer fly. Natural selection initially favored this because, without predators, there is no point using valuable food to grow powerful wings. However, if predators do eventually arrive, then the birds will have no escape. Such an evolutionary “mistake” probably explains the evolution of balanced lethal systems, but we do not yet know exactly what happenend [5]. In the two centuries since the bizarre phenomenon of balanced lethal systems was first discovered in crested newts, we have learned a lot. But, so far, scientists have only studied the chromosomes. With modern techniques, it is possible to take a much closer look at the DNA code inside the chromosomes themselves. Scientists are now planning to study the crested newt’s chromosome 1 in this way. Surely, the DNA code must hold the key to solving the mystery of balanced lethal system evolution!
Glossary
Embryo: ↑ A yet unhatched or unborn individual that is developing inside an egg or uterus.
Evolution: ↑ The change of traits within a species, encoded in their DNA, over successive generations.
Chromosome: ↑ Long chains of densely packed DNA that are present in every cell and together contain an organism’s genetic code.
Recombination: ↑ The step during the production of sperm or egg cells in which chromosome pairs, with one member each from an individual’s mother and father, swap DNA and are divided.
Balanced Lethal System: ↑ A situation where two versions of a chromosome exist that are both required for survival; every generation half the offspring receive two copies of the same version and dies.
Natural Selection: ↑ The evolutionary process by which individuals that best suit the environment due to favorable traits are more likely to survive and have offspring that inherit these same traits.
Adaptation: ↑ A trait, resulting from and maintained by evolution by natural selection, that makes an individual suited to deal with a particular environment.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We thank Michael Fahrbach for allowing us to use his newt pictures in the figures. This project has received funding from the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation programme (Grant Agreement No. 802759). The PhD position of WM was supported by the Nederlandse organisatie voor Wetenschappelijk Onderzoek (NWO Promotiebeurs voor leraren 023.016.006).
References
[1] ↑ Rusconi, M. 1821. Amours des salamandres aquatiques: et developpement du tetard de ces salamandres depuis l’oeuf jusqu’a l’animal parfait. Milano: Giusti.
[2] ↑ Macgregor, H. C. 1991. Chapter 8 - Chromosome heteromorphism in newts (Triturus) and its significance in relation to evolution and development. In: S. K. Sessions, editor. Amphibian Cytogenetics and Evolution. San Diego, CA: Academic Press. p. 175–96.
[3] ↑ Macgregor, H. C., and Horner, H. 1980. Heteromorphism for chromosome 1, a requirement for normal development in crested newts. Chromosoma. 76:111–22. doi: 10.1007/BF00293412
[4] ↑ Wielstra, B. 2020. Balanced lethal systems. Curr. Biol. 30:R742–3. doi: 10.1016/j.cub.2020.05.011
[5] ↑ Grossen, C., Neuenschwander, S., and Perrin, N. 2012. The balanced lethal system of crested newts: a ghost of sex chromosomes past? Am Nat. 180:E174–83. doi: 10.1086/668076
