Abstract
Bacteria live with each other in an organized network called a biofilm. The biofilm allows the bacteria to communicate with each other and transfer nutrients and signals from one bacterium to another. This communication provides the bacteria with new properties that allow them to survive, even when they are exposed to harmful compounds that usually kill them (for example, antibiotics). These persistent bacteria can now cause what are called hospital-acquired infections because patients often get them while in a healthcare facility, like a hospital. Hospital-acquired infections obviously endanger the health of patients and may lead to death. So, it is important to find solutions that prevent bacteria from forming biofilms. My research group designed and synthesized a compound that forms a coating on different materials and does not allow bacteria to form biofilm. This compound may be useful in the future as a coating for medical devices, water desalination facilities, and food preparation surfaces.
What are Bacteria and How Do They Like to Live?
Bacteria are small organisms that cannot be seen with the naked eye. To detect them, one must use a microscope. A bacterium is made up of a cell wall that surrounds the genetic material and other structures that are needed for the bacterial cell. Bacteria are the most abundant form of life on our planet. They exist in multiple sizes, shapes, and colors. Bacteria reproduce by division, where each cell divides into two new cells, called daughter cells. Within half an hour or so, bacteria can double in number. Many bacteria are not harmful to humans and are considered “good” bacteria. These good bacteria are beneficial to humans. For example, “good” bacteria can take up the space of harmful bacteria and by doing so protect us from infections. Other bacteria help us to digest our food [1]. Harmful bacteria, also called pathogenic bacteria, cause various illnesses.
Bacteria live in colonies, in which each bacterium is surrounded by many other bacteria that protect it from the external environment. Within the colony, the bacteria communicate with each other using chemicals. These chemical signals help the bacteria to form a network called a biofilm (Figure 1). Another name for a biofilm is a plaque. The term plaque is often used by dentists to describe the bacterial layer on teeth. This layer is well attached to the teeth and secretes materials that destroy the teeth. A biofilm can provide bacteria with resistance to antibiotics, which can usually kill individual bacteria (Figure 1). As a biofilm, some bacteria can still live, multiply, and cause infections—even in the presence of antibiotics. These infections are a major problem in hospitals, where they harm and even kill the patients. To prevent these infections, hospitals and the healthcare system invest much effort to clean hospital facilities and medical devices [2].
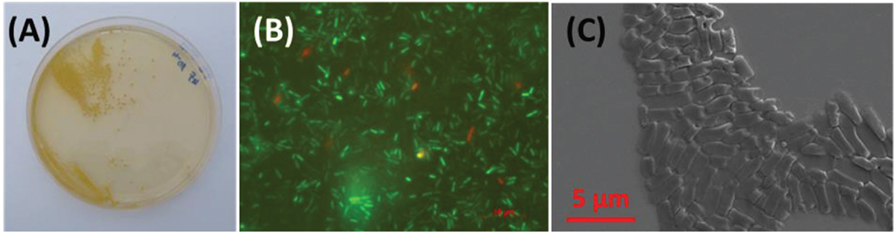
- Figure 1
- What do bacteria look like? A. Bacteria live together and form colonies on the surface of the laboratory dish. In the picture, the colonies are the round yellow dots. Each dot contains thousands of bacteria. B. It is possible to dye the bacteria with a fluorescent dye that colors the live cells green. This staining method allows us to test materials for their antimicrobial activity. C. Each colony is made up of many micron-size bacteria. A micron, also called a micrometer (μ) is 10−6 m.
How Do Antibiotics Work?
An antibiotic is a compound that kills bacteria. Antibiotics stop essential cell activities that allow the bacteria to live. For example, some antibiotics harm the cell wall and some prevent the bacteria from reproducing. The first antibiotic, penicillin, was discovered over 90 years ago. Since then, many other antibiotics have been found. Since the discovery and use of antibiotics, bacteria have evolved that resist antibiotics and multiply even when antibiotics are present. These are called “antibiotic-resistant bacteria.” Antibiotic resistance evolves by mutations (genetic changes) in the bacterial DNA that allow the bacteria to survive in the presence of antibiotics. A year ago, the World Health Organization published a report on 12 different bacterial strains that are resistant to antibiotics [3]. For these antibiotic-resistant bacterial strains, solutions other than antibiotics must be found to kill the bacteria. My research group believes that, if we can prevent biofilm formation, we will be able to successfully fight the antibiotic-resistant bacteria.
How Do We Fight Bacteria?
Many research groups around the world are trying to find compounds that will prevent biofilm formation. Some of that research focuses on finding compounds that will prevent the first step of biofilm formation. In the first step of biofilm formation, large molecules such as proteins and polysaccharides (large sugars) are produced by the bacteria and stick to the surface that the bacteria are growing on. These substances provide a “glue” for bacteria, and that is how biofilm formation starts.
Over a decade ago, several research groups showed that small particles of silver can kill bacteria and the groups suggested that these particles might be able to act as a new antibiotic. However, it was later found that these particles can also kill human cells and are dangerous to humans. So, silver particles were banned from use. Another material that was suggested to possibly prevent biofilm formation was polyethylene glycol (PEG). This compound can bind up many water molecules, which prevents the bacteria from sticking to the surface. However, PEG breaks down over time, and so it is not possible to use it for long-term applications. Other researches tried to mimic natural surfaces that prevent bacteria from sticking to them. For example, lotus leaves are always clean, even though this plant usually grows in swamps and shallow water. The lotus leaves stay clean because there are many bumps on the leaves that are covered with wax. The combination of bumps and wax helps a water drop to slip from the leaf, dragging all the dirt with it, like a mop. This phenomenon is called “the lotus effect.” Many research groups are trying and even succeeding in mimicking this effect. However, mimicking the lotus effect often requires expensive and complicated production processes that are limited to small surfaces. Therefore, this solution cannot yet be used in hospitals or other places where we want to prevent biofilm formation.
Interrupting the communication among bacteria is another possible way to prevent biofilm formation. Bacteria communicate with each other using various chemicals. One bacterium produces a certain molecule and another receives it and produces a different molecule as a response. Synthesizing molecules that harm this communication, by blocking the signals, is another option to fight biofilm formation [4].
Our Proposed Solution
My research group developed a small molecule that prevents the sticky biological molecules (proteins, sugars, etc.) from adhering to surfaces, and therefore reduces biofilm formation [5]. To form this “non-stick” coating that will prevent the attachment of biological molecules to a surface, we mimicked the chemistry of Teflon®. Teflon® is a non-stick material used in the production of frying pans. This molecule we developed is made up of amino acids. Amino acids are the building blocks of proteins. We chose amino acids because they are not toxic to humans, they are easy to synthesize in large quantities, they can be stored in the refrigerator for years, and, most interestingly, because they can self-assemble. Self-assembly is a process by which molecules spontaneously form an ordered structure, without any need for external energy. In other words, these amino acids can interact with each other and connect like Lego building blocks.
To adhere our self-assembling non-stick molecules to the surface, we used another amino acid, one that serves as the main component of mussel’s adhesive proteins (MAPs). Mussels can attach strongly to rocks in the sea and hang on during high and low tide. Mussels adhere using strings made of MAPs that are rich in the amino acid L-3,4-dihydroxyphenylalanine (DOPA). This amino acid enables MAPs to adhere to any surface—metal, glass, and plastic.
We synthesized our non-stick molecule in the lab, in the form of a powder that looked like powdered sugar. We dissolved this powder in a liquid and sprayed it on different surfaces. We then performed several measurements to prove that the molecules indeed self-assembled on the surface. To test if the coating prevented bacterial adhesion, we incubated the coated surface in a solution that contained a million bacterial cells and their required nutrients. We always tested two types of surfaces: one with the coating (the experiment) and the other without the coating (the control). The surface without the coating served as a control, because it showed us the number of bacteria that would adhere to a surface without a coating. According to our previous experiments, the strain of bacteria that we used produced a biofilm on the surface within 9 h. Therefore, we waited 9 h before we took the surfaces out of the solution of bacteria. To determine how many bacteria we had on the surface, we scratched the surface with a toothbrush and transferred the bacteria from the toothbrush to a plate that contained food for the bacteria. This plate is called a Petri dish (Figure 2). Bacteria grew on the Petri dish and formed colonies. Each colony resulted from one bacterium that came off the surface. We counted the number of colonies and found that the number of bacteria on the coated surface was significantly smaller than the number of bacteria on the non-coated surface.
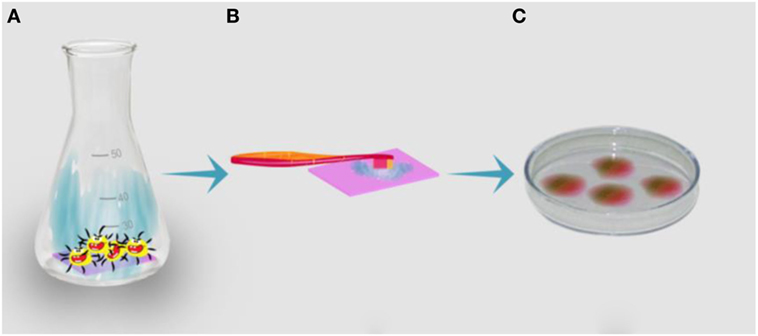
- Figure 2
- Determining the number of bacteria on a surface. A. In the first step, the surface (the colored rectangle at the bottom of the container) is placed in a container that holds the bacteria and their nutrients. B. In the second step, the surface is taken out of the container and the bacteria are scraped from the surface using a toothbrush. C. During the third step, the bacteria are plated on a dish that contains all the necessary food for the bacteria. On this solid surface, the bacteria grow into colonies. The colonies appear as rounded spots or dots on the plate (see Figure 1A). Each colony originates from one bacterium, and therefore, the number of colonies reflects the number of bacteria on the original surface.
To make this coating useful in real life applications, we still need to prove that the coating can reduce the number of bacteria under normal conditions in the outside environment. This means that the coating should survive sunlight, scratches, and exposure to high and low temperatures. In addition, it is necessary to prove that the material is not toxic to humans. These tests are called regulation assays, and they are required for any new material. The material we developed is currently being tested in regulation assays.
What about the Future?
Unfortunately, an ultimate solution that completely prevents biofilm formation does not exist. Biofilms not only cause hospital-acquired infections but they also harm the quality of food and water. Research groups that try to solve the problem of biofilms have come up with several different solutions, but there is still a need for a coating that will be stable enough, will not come off from the surface, will not be toxic to cells, and will be easy to apply and inexpensive. We hope that the solution we proposed, made up of amino acids, will provide a good solution to the problem. Do you have your own idea for a solution to this problem?
Glossary
Cell Wall: ↑ A rigid layer made of proteins and sugars that defines the borders of the cell, provides it with physical support, and protects it from the environment.
Cell: ↑ The most basic living unit that has all the characteristics of life—respiration, motion, and reproduction.
Molecule: ↑ A substance that is made up of more than two atoms that are connected by a chemical bond.
Amino Acids: ↑ The molecules that make up proteins.
Conflict of Interest Statement
The author declares that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
[1] ↑ Black, J. G. and Black, J. L. 2015. Microbiology: Principles and Explorations. 9th ed. Hoboken, NJ: ohn Wiley & Sons, Inc.
[2] ↑ Percival, S. L., Suleman, L., Vuotto, C., and Donelli, G. 2015. Healthcare-associated infections, medical devices and biofilms: risk, tolerance and control. J. Med. Microbiol. 64:323–34. doi:10.1099/jmm.0.000032
[3] ↑ WHO. 2017. WHO Publishes List of Bacteria for Which New Antibiotics are Urgently Needed. GENEVA: WHO. Available at: http://www.who.int/medicines/publications/global-priority-list-antibiotic-resistant-bacteria/en/
[4] ↑ Nir, S., and Reches, M. 2015. Bio-inspired antifouling approaches: the quest towards non-toxic and non-biocidal materials. Curr. Opin. Biotechnol. 39:48–55. doi:10.1016/j.copbio.2015.12.012
[5] ↑ Maity, S., Nir, S., Zada, T., and Reches, M. 2014. Self-assembly of a tripeptide into a functional coating that resists fouling. Chem. Commun. 50:11154–7. doi:10.1039/c4cc03578j