Some patients with neurological diseases (e.g., a disease that involves abnormal brain function) do not respond well to the available medications and must resort to alternative surgical therapies to manage their symptoms. Parkinson's disease (PD), for example, involves damage to a specific brain area called the basal ganglia and is characterized by reduced levels of a substance called dopamine in the brain. These changes to the brain physiology are associated with motor symptoms like imbalance, tremor, and slowness of motion, which can be very disabling. Fortunately, medications like levodopa can partially return the balance of dopamine in the brain and relieve the motor symptoms of the disease, especially in its early stages. However, as PD progresses, the drug therapy becomes less and less effective, and an additional therapy is needed.
Electrical stimulation of the basal ganglia [e.g., the same structure that demonstrates abnormal physiology in Parkinson's disease (PD) patients] relieves many of the motor symptoms of the disease. The treatment involves the placement of a permanent electrode in the basal ganglia as well as of a brain pacemaker that is placed under the skin of the chest and provides the power needed to produce electrical stimulation to the electrode inside the brain (Figure 1). Typically, the electrode incorporates four contacts that can provide various types of electrical stimuli. Therefore, there are many ways in which a physician can stimulate the brain using one electrode. The effect of the electrical stimulation on the motor symptoms of PD is almost immediate. Involuntary hand tremors can stop at once, the patients often get better control of their gait, and their quality of life improves. Today, deep brain stimulation for PD, and for other neurological movement disorders, is a routine procedure with proven benefits. Moreover, its applications for additional neurological and psychiatric disorders are under extensive investigation with promising results for depression, obsessive-compulsive disorders, and epilepsy, among others.
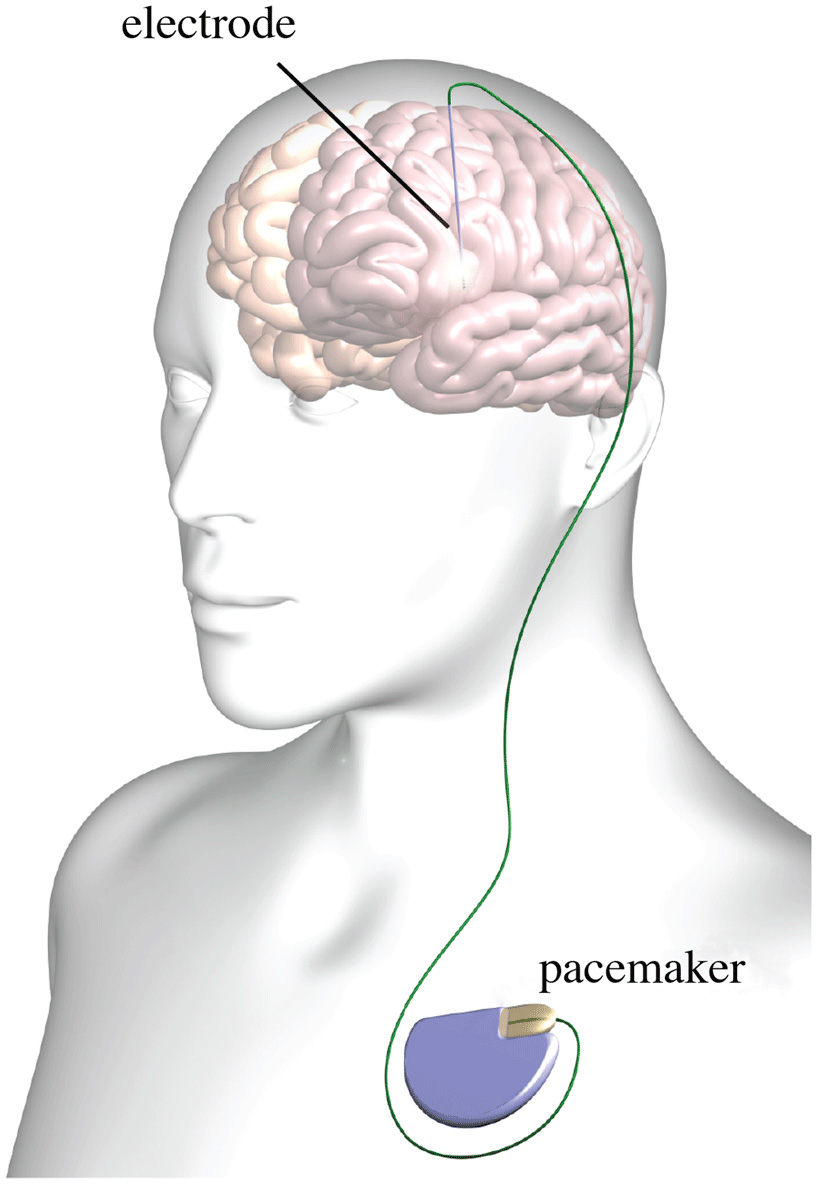
- Figure 1 - Typical deep brain stimulation setup.
- The electrode is placed in the brain and connected to a brain pacemaker permanently placed under the skin of the chest.
Despite its effectiveness, the outcomes of the deep brain stimulation treatment may vary with time and among patients, and adverse effects may appear. Therefore, scientists and physicians put much effort into improving the treatment's outcomes and avoiding side effects. General guidelines for patient selection and careful treatment directions were formulated for the safe treatment of PD and other movement disorders, based on clinical data gathered in the past decade. Apparently, the general guidelines may prohibit an effective treatment from PD patients and it was estimated that about one out of three patients that would benefit from deep brain stimulation are excluded if strictly following the guidelines. Moreover, the guidelines cannot cover the large variability among patients, and two patients with different symptoms can receive the same treatment, which may be less effective compared to a treatment that is tailored for the specific patient. Therefore, the focus of our research has moved to a patient-specific approach, where the treatment is tailored to the patient's specific symptoms and clinical history, among other factors.
Traditionally, it was believed that electrical stimulation deactivates the effected target brain area. However, recent studies suggest that the effect of this treatment is more complex, with the deactivation of some zones and the activation of others. Our team computes for each patient the spread of electrical stimulation in the brain, estimates the neural zones that are activated by a specific electrical stimulation, and relates the activated neural zones to the actual PD treatment outcomes and adverse effects (see Figure 2). A physician would be able to test multiple electrical stimulation parameters (e.g., voltage, frequency, etc.) and get a good estimate of their outcomes and adverse effects in a short time and without the patient attendance. A custom module would be able to automatically compute the best electrical stimulation setup to maximize treatment effectiveness and shorten office visit time. This optimization would be with respect to the patient-specific symptoms and medications. For example, the optimal stimulation for a patient with left hand tremor could be different from that of a patient with left leg slowness. We hope to apply this approach to other diseases in the near future.
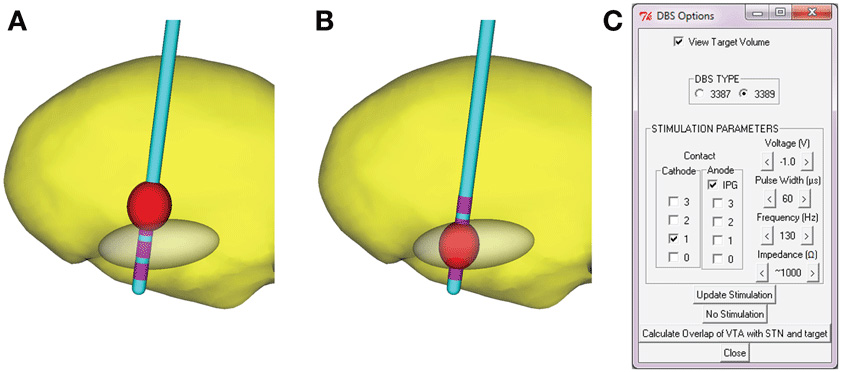
- Figure 2 - A model for the effect of electrical stimulation in a patient's brain.
- The red volume represents the brain volume that will become activated under the specific electrical stimuli that is being considered. A stimulation of the gray area is expected to result in the most desired outcomes. Therefore, the greater the overlap between the red and gray volumes, the better. In our illustration, the stimulation setup that activates the brain tissue inside the gray zone (B) is preferred to the stimulation setup that is activating brain tissue mostly outside the gray zone (A). Image (C) shows the many variables that affect the stimulation results. Imagine how hard it is to decide on the values that will relieve the patient's symptoms without any guidance.
Another interesting approach is to record the patient's brain activity under motor, cognitive (e.g., memory, language, etc.), and psychiatric activities, and to create a map that relates function to tested electrode locations. Then, the electrode is placed in such a way that the target area receives most of the stimulation. The timing of stimulation is also under study. Scientists suggested stimulating only when abnormal brain activity is present and observed some benefits in animal models of PD. Further studies are needed to better understand its full range of effects.
In summary, deep brain electrical stimulation can be used to relieve neurological and psychiatric diseases that would otherwise remain untreated. The focus of our research today is on a patient-specific approach, for best treatment outcomes and minimal adverse effects.
References
[1] ↑ Butson, C. R., Cooper, S. E., Henderson, J. M., Wolgamuth, B., and McIntyre, C. C. 2011. Probabilistic analysis of activation volumes generated during deep brain stimulation. Neuroimage 54:2096–104. doi: 10.1016/j.neuroimage.2010.10.059
[2] ↑ Eitan, R., Shamir, R. R., Linetsky, E., Rosenbluh, O., Moshel, S., Ben-Hur, T., et al. 2013. Asymmetric right/left encoding of emotions in the human subthalamic nucleus. Front. Syst. Neurosci. 29:69. doi: 10.3389/fnsys.2013.00069
[3] ↑ Frankemolle, A. M., Wu, J., Noecker, A. M., Voelcker-Rehage, C., Ho, J. C., Vitek, J. L., et al. 2010. Reversing cognitive-motor impairments in Parkinson's disease patients using a computational modelling approach to deep brain stimulation programming. Brain 133:746–61. doi: 10.1093/brain/awp315
[4] ↑ Rosin, B., Slovik, M., Mitelman, R., Rivlin-Etzion, M., Haber, S. N., Israel, Z., et al. 2011. Closed-loop deep brain stimulation is superior in ameliorating parkinsonism. Neuron 72:370–84. doi: 10.1016/j.neuron.2011.08.023
[5] ↑ Starr, P. A., Vitek, J. L., and Bakay, R. A. 1998. Ablative surgery and deep brain stimulation for Parkinson's disease. Neurosurgery 43:989–1013. doi: 10.1097/00006123-199811000-00001