Abstract
Brain tumors are abnormal growths of cells in the brain, and they can be cancerous or non-cancerous. My job as a neurosurgeon is to perform surgeries to remove harmful brain tumors from patients’ brains. Along with this work, I am also a professor at the University of Toronto, where I study brain tumors. By learning more about brain tumors in the lab, I hope to help develop new treatments and diagnostic tools that can improve the ways patients are treated and the outcomes of those treatments. In this article, I will tell you about brain tumors, how they are usually diagnosed and treated, and the next steps we are hoping to take to improve the field of neurosurgery.
Dr. Gelareh Zadeh won the 2023 Canada Gairdner Momentum Award for advancing our understanding of brain tumors at the molecular level. Her research is leading to better ways of identifying, classifying, and managing different types of brain tumor. This has the potential to improve the medical care that doctors provide to patients suffering from brain tumors.
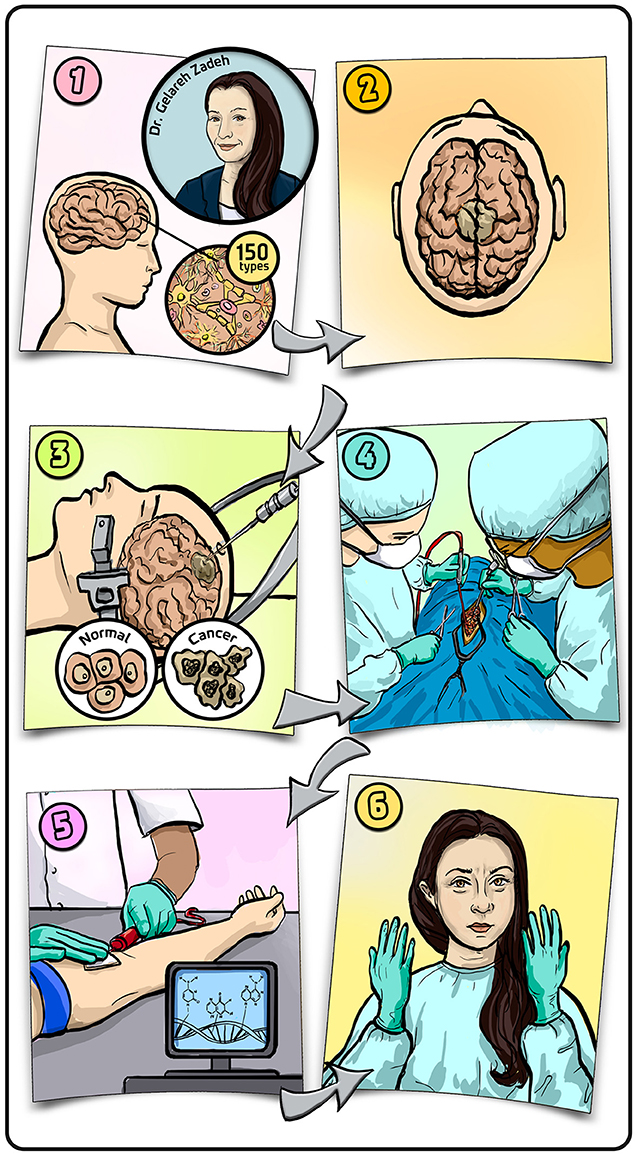
- Graphical Abstract - (1) There are more than 150 types of brain tumors, defined by the types of cells they arise from and whether they are cancerous or not.
- (2) A tumor in the brain looks like a lump of cells that have grown abnormally. (3) To determine whether a tumor is cancerous, patients undergo a biopsy in which part of the tissue is surgically removed and analyzed in the lab. (4) If the tumor is cancerous, patients have surgery to remove as much of the tumor as possible. (5) We hope to replace biopsies with blood tests that can diagnose the type of tumor each patient has. (6) What I love about being a neurosurgeon is how my brain gets into full concentration mode from the second I put on my gloves. Illustration by: Iris Gat.
What Are Brain Tumors?
The brain is a very valuable organ. In many ways, the brain makes us who we are, with our unique personalities and behaviors. The brain is also very sensitive, and its functioning can be altered by several factors. Brain tumors, which are abnormal growths of cells in the brain, are one of the things that can alter brain functioning. There are over 150 different types of brain tumors [1]. The brain is made of many types of cells, and brain tumors can come from any of these cell types. Some brain tumors are benign, meaning that they are not cancerous and do not spread inside the brain. Benign brain tumors are typically cells that have grown out of the normal pattern and, as a result, they put pressure on the brain area where they grow. Some tumors are malignant (cancerous) and they can grow and spread rapidly in the brain.
Most neurosurgeon, myself included, usually manage and deal with only about eight to ten common types of brain tumors on a regular basis. Some of the most common brain tumors are glioblastomas and gliomas, which are aggressive cancerous growths; meningiomas, which are generally benign tumors that form from the covering of the brain; and pituitary tumors, which are non-cancerous and form from cells of the pituitary gland, but can compress the nerves connecting the eyes to the brain and cause vision problems. The most challenging tumors to handle are generally those that are wrapped around nerves or blood vessels. In trying to remove such tumors, neurosurgeons can potentially harm the functioning of the nerves or create bleeding in the brain by wounding the blood vessels. These tumors require extremely delicate removal with the use of a surgical microscope. Tumors that are not wrapped around nerves or blood vessels can usually be successfully removed by surgery.
How Are Brain Tumors Diagnosed and Treated?
Patients with brain tumors usually arrive at the hospital experiencing problems with some brain function (Figure 1A), such as a problem with eyesight or hearing, memory, or movement of the limbs; or they might be having seizures. Doctors normally do brain scans on these patients. A common scan is called a computed tomography (CT) scan, which is based on X-rays. If the CT scan finds something suspicious, doctors can also do a more detailed scan using a technique called magnetic resonance imaging (MRI) (Figure 1B). Using the MRI scan, neurosurgeons can confirm whether there is a brain tumor and sometimes tell what type it is. To be sure of the diagnosis, patients often have a biopsy, in which part of the suspected tumor is surgically removed and analyzed in a pathology lab (Figure 1C).
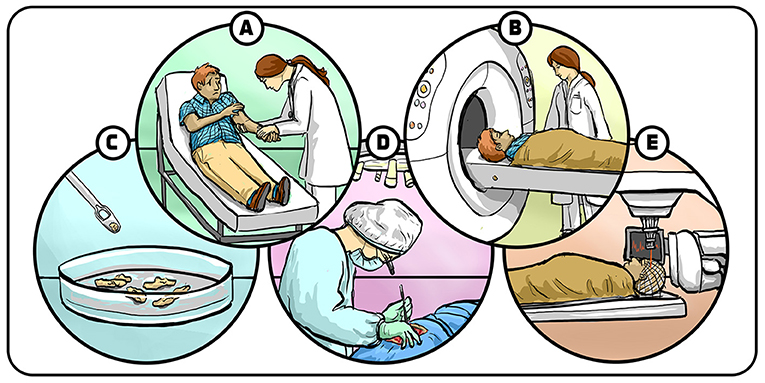
- Figure 1 - Diagnosis and treatment of brain tumors.
- (A) Patients with brain tumors often arrive in the hospital complaining about a brain-related problem, such as trouble moving their arms or legs. (B) Patients are sent for a brain scan and (C) a biopsy, to identify whether they have a tumor and they type of tumor. (D) If the tumor is cancerous, it is often removed by surgery, followed by (E) radiation treatments and chemotherapy, which target the parts of the tumor that could not be removed by surgery. Illustration by: Iris Gat.
In the case of benign tumors that do not involve critical brain structures like nerve cells and blood vessels, the tumors are removed and usually do not come back. In the case of cancer, the situation is more complex. Brain cancers spread into different tissues in the brain and commonly cannot be removed completely. The parts that can be safely removed are removed surgically (Figure 1D), and the rest of the tumor is treated with other therapies, such as radiation and chemotherapy, to try to shrink or eliminate the remaining tumor (Figure 1E). Despite the best efforts of neurosurgeons and cancer doctors, sometimes brain tumors come back. Currently, about 30% of the patients in my clinic have repeated surgeries. We can do more than one surgery because our modern surgical techniques and technologies are precise, effective, and do not cause a lot of damage to patients’ brains, so the recovery process is easier for patients than it used to be. Part of my motivation as a neurosurgeon is to improve the outcomes of surgeries and find treatments that reduce the negative effects of surgery, radiation, and chemotherapy.
Can We Use Blood Tests to Diagnose Brain Cancer?
Today, brain cancers are diagnosed using biopsies. Brain biopsies require cutting into the brain, which can be risky and anxiety provoking for the patients, so we would like to find an easier but at the same time reliable way. It may be possible to use blood tests to diagnose brain cancers [2]. These blood tests are based on biomarkers, which are biological materials that are present in the blood and that we can measure. Tumors often release biomarkers into the blood by shedding parts of themselves, including their DNA. We can capture and concentrate tumor DNA present in the blood using magnetic methods, and analyze its composition. When we analyze tumor DNA, we are looking for a specific “signature” of molecules containing three hydrogen atoms and one carbon atom. Molecules of this form are called a methyl group, and they get attached to different parts of the DNA. These methyl groups are like markers signaling which genes should be silenced and which genes should be activated. This process of silencing and activating genes using methyl groups is called DNA methylation [3]. DNA Methylation is unique to each tissue, so it can serve like a specific “fingerprint” of that tissue. In our case, methylation can serve as a fingerprint for each of the 150 types of brain tumors. If we do a blood test and find DNA that has the methylation signature of a certain type of brain tumor (Figure 2), we can in principle know which type of tumor the patient has without having to take a sample of the tumor through a biopsy.
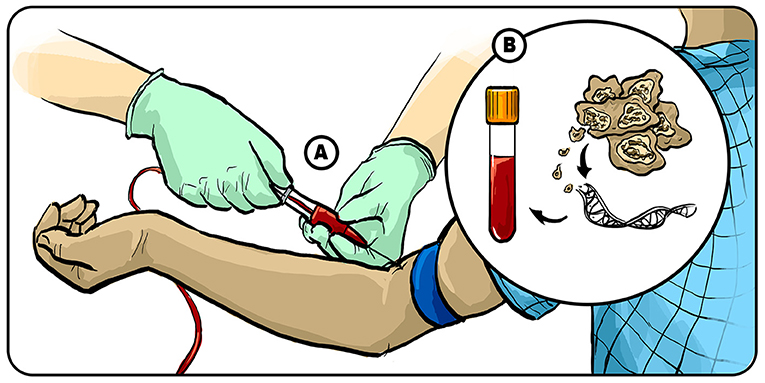
- Figure 2 - Using blood tests to diagnose brain tumors.
- (A) We are currently working on developing blood tests to diagnose which type of brain tumor a patient is suffering from. (B) After we take the patient’s blood, we concentrate the DNA biomarker from the tumor and look at the DNA methylation “fingerprint”, which can tell us which type of brain tumor the DNA came from. Illustration by: Iris Gat.
Currently, the accuracy of blood tests to diagnose brain cancers is about 80%. Before blood tests can replace biopsies, we must increase the accuracy to at least 90%. In medicine, new treatments must surpass existing treatments before they become the new standard. Even after a new standard is reached, it usually takes a few years until it is widely accepted by doctors around the world. I believe that, in a few years, diagnostic blood tests for brain tumors will replace biopsies. That will revolutionize the way we treat brain tumors—not only because we will no longer need biopsies, but also because blood tests could be a great tool for detecting tumors that return after surgery. Currently, we rely on MRI to see if tumors come back after surgery, but by the time we see the tumor using MRI, the tumor has already grown quite large. Our aim is to have sensitive blood tests for DNA methylation, to diagnose a returning cancer long before it reaches a critical mass of cancerous cells. This could help us treat returning cancers early, which could save lives. Additionally, when tumors come back, they often have a different composition of cells, depending on which cells survived previous treatments. In other words, the cells that survived may have a different genetic mix than the original tumor. This means that the second treatment might need to be different than the first—and knowing the exact composition of the returning tumor based on its methylation signature could help us design the best treatment.
The Future of Brain Cancer Therapy
Over the last few decades, our ability to identify tumors based on DNA biomarkers has significantly increased. Now we know that specific methylation signatures are associated with specific tumors, which tells us that there are many subtypes of the types of brain tumors we previously identified. Tumors that look identical under the microscope may be classified differently based on their genetic mutations, and the precision with which we are able to recognize these tumors using blood samples is increasing. The next step is to develop specific treatments for each type of tumor.
We recently published a study in which we used DNA differences to predict how effective a treatment would be for a brain tumor called glioblastoma [4]. We injected a virus called adenovirus into the glioblastoma tumor. When the virus enters cancerous cells, it replicates inside the cells and the cells burst. This burst triggers the immune system to attack cancerous cells. We also gave the patients medicines to boost their immune systems. We looked both at the tumors and at the immune cells to see if there was a genetic signature that could tell us which patients would respond well to this treatment and which would not. We found that, if certain genes of the immune system were highly activated, then patients were likely to respond to the treatment. If the genes were minimally activated, it was not likely that the patient would respond well to the treatment. This is an important demonstration of our ability to use genetic signatures to “tailor” treatments to specific patients. We hope that future studies like this one will help us to understand which treatments our patients should respond to. We also want to develop more treatments for brain tumors.
I have dedicated my life to both researching how to improve diagnosis and treatments for brain tumors, and to performing brain surgeries on patients. Learning how to balance research and medical practice was a big challenge. Another challenge that I had to face was how to avoid becoming discouraged when complications happen to patients. As a doctor, I naturally want all my surgeries to be perfect—but sometimes things get complicated, and patients are left in challenging situations. This is very tough for any neurosurgeon to experience. I found that talking to with honesty to patients and colleagues about these experiences, while also learning how to manage continuing to take care of the patients is very important and helpful. I also make sure to learn from every experience I have by giving my fullest attention to what I do—be it caring for patients or conducting scientific research.
Glossary
Benign: ↑ Non-cancerous tumors that do not spread to other parts of the body.
Malignant: ↑ Cancerous tumors that can spread to other parts of the body.
Neurosurgeon: ↑ A surgeon that specializes in surgeries on the nervous system, especially the brain and spinal cord.
Biopsy: ↑ A surgical procedure in which part of a suspicious tissue is removed to be inspected in the lab.
Radiation: ↑ A therapy that uses very energetic beams (commonly x-rays) to kill or slow down the growth of cancerous cells.
Chemotherapy: ↑ A therapy that uses strong drugs to harm cancerous cells by disrupting their growth and division processes of cancerous cells.
Biomarker: ↑ A biological material that can be found in body fluids like blood and is used to diagnose a disease.
DNA Methylation: ↑ A way in which the body turns genes on and off, by adding or removing chemical groups called methyl groups to the DNA.
Conflict of Interest
The author declares that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional Materials
- Dr. Gelareh Zadeh-2023 Canada Gairdner Momentum Award.
- Meet 2023 Gairdner Momentum Award winner Dr. Gelareh Zadeh (YouTube).
Acknowledgments
I wish to thank Noa Segev for conducting the interview which served as the basis for this paper and for co-authoring the paper, and Iris Gat for providing the figures.
References
[1] ↑ Kleihues, P., Louis, D. N., Scheithauer, B. W., Rorke, L. B., Reifenberger, G., Burger, P. C., et al. 2002. The WHO classification of tumors of the nervous system. J. Neuropathol. Exper. Neurol. 61, 215–225. doi: 10.1093/jnen/61.3.215
[2] ↑ Nassiri, F., Chakravarthy, A., Feng, S., Shen, S. Y., Nejad, R., Zuccato, J. A., et al. 2020. Detection and discrimination of intracranial tumors using plasma cell-free DNA methylomes. Nat. Med. 26, 1044–1047. doi: 10.1038/s41591-020-0932-2
[3] ↑ Moore, L. D., Le, T., and Fan, G. 2013. DNA methylation and its basic function. Neuropsychopharmacology 38, 23–38. doi: 10.1038/npp.2012.112
[4] ↑ Nassiri, F., Patil, V., Yefet, L. S., Singh, O., Liu, J., Dang, R. M., et al. 2023. Oncolytic DNX-2401 virotherapy plus pembrolizumab in recurrent glioblastoma: a phase 1/2 trial. Nat. Med. 29, 1370–1378. doi: 10.1038/s41591-023-02347-y
